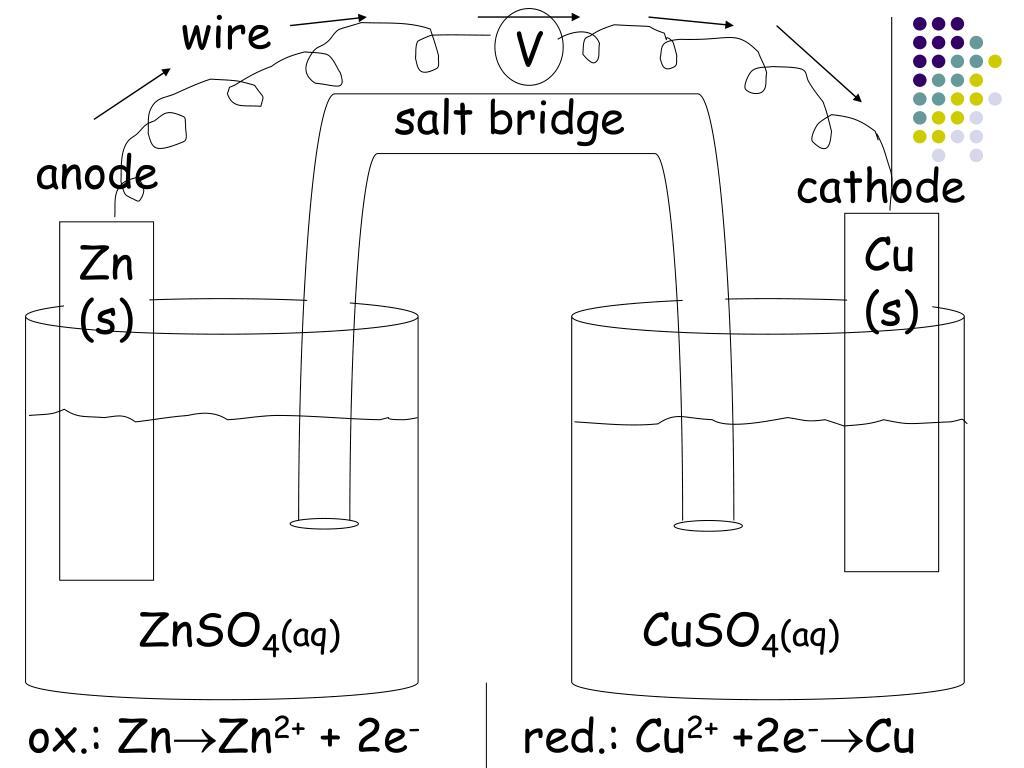
Why don't Zn or Cu ions flow through the salt bridge and end up on each side? Remember ions can only flow in the solution they are in (usually water or some kind of liquid) For a Zn ion to flow across the salt bridge it would have to first defy gravity to get up the first part of the bridge, resist bonding with sulfate ions, and continue all the way across the other side to the copper solution. The salt bridge now allows for Na+ ions to flow to the right side to balance out the negative charge and keep the battery working. Now however our right side is becoming to negatively charged and starting to repel electron flow. Once the Zn ion solution becomes too positive the salt bridge allows negative Sulfate ions to flow into the zinc side. Our battery stops working!Ī salt bridge however balances the charges of the solutions on both sides. What would happen then? No electron flow = no current. As it becomes more and more positive electrons can no longer travel to the copper because they are attracted to this strong positive charge. Zinc ions in the solution are positive, and hence the solution on the left side becomes more positive. The zinc half-cell acts as the anode.Here's a more in depth look at what's happening:Įlectrons are flowing from the zinc solid metal strip to the copper strip. Zinc is higher up on series than copper thus it is more easily oxidized. To determine which metal will be oxidized, or which will be reduced, refer to the activity series.The high concentration allows ions to diffuse out of it. Salt bridge contains a concentrated solution of a strong electrolyte.This is the voltage generated when two different solutions come into contact with each other Reduces the liquid-junction potential.Provides electrical continuity for anions and cations.Provides physical separation of reduction and oxidation processes.Salt bridge which connects both electrodes has multiple functions:.The vertical line represents a phase boundary (junction).Consists of a bar of metal placed in a solution containing cations of the same metal.There are different types of electrodes used in voltaic cells, most common one is metal/metal ion electrode.Oxidation occurs at one half-cell (anode), reduction occurs at on half-cell (cathode) These two are separated, connected only by a salt bride. The salt bridge is represented by two parallel lines By convention, anode is always of left, and cathode on right. Cell Diagrams are used as shorthand notation to represent a voltaic cell.In both voltaic and electrolytic cells:.Electrochemical cells contain two electrodes, anode and the cathode.An electrode is a conductor of electricity used to make contact with a non-metallic part of a circuit, such as the solution in a cell (electrolyte).Electrolytic Cells – convert electrical to chemical energy.Reactions are spontaneous and exothermic. Voltaic (Galvanic) Cells – convert chemical to electrical energy.There are two main types of electrochemical cells.In an electrochemical cell, there can either be a conversion of chemical energy to electrical energy, or the other way around.D.7 (HL) Taxol – a chiral auxiliary case study.D.6 Environmental impact of some medications.D.1 Pharmaceutical products and drug action.C.8 Photovoltaic and dye-sensitized solar cells.C.6 Electrochemistry, rechargeable batteries and fuel cells.C.5 Environmental impact – global warming.A.10 Environmental impact – heavy metals.A.8 Superconducting metals and X-ray crystallography.A.2 Metals and inductively coupled plasma (ICP) spectroscopy.21.1 Spectroscopic identification of organic compounds.18.2 Calculations involving acids and bases.16.1 Rate expression and reaction mechanism.14.1 Covalent bonding and electron domain and molecular geometrics.The periodic table – the transition metals 11.3 Spectroscopic identification of organic compounds. 
11.1 Uncertainties and errors in measurements and results.6.1 Collision theory and rates of reaction.1.1 Introduction to the particulate nature of matter and chemical change.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
